Separation Science Laboratory > Research > Research Field
Research Field
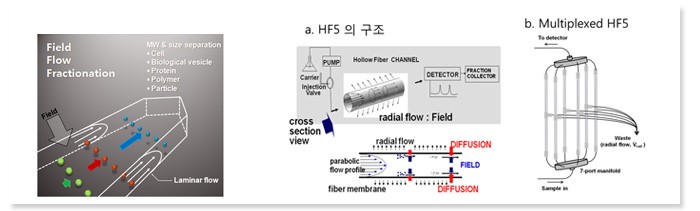
Our research has been focused to develop analytical methods to analyze new particulate materials, pharmaceutical macromolecules, proteins, and lipids by combining Field-Flow Fractionzation (FFF), Nanoflow Liquid Chromatography (nLC), Multiangle Light Scattering (MALS), and Electrospray Ionization Mass Spectrometry (ESI-MS)Flow Field-Flow Fractionation (FIFFF)


Among different techniques of FFF, our research focuses on optimizing and improving Flow Field-Flow Fractionation (FlFFF) techniques to analyze biological materials and nanoparticles.
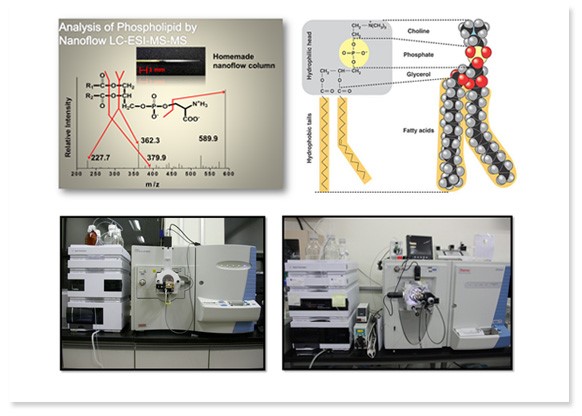
Nanoflow LC-ESI-MS/MS for proteomics and lipidomics

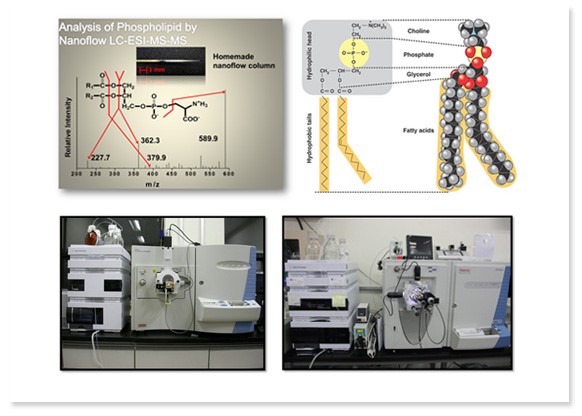 Metabolites are directly or indirectly responsible for metabolic process, growth, and development, and affected by changes in external environments unlike DNA or proteins. They are mainly composed of lipids which are defined to be insoluble in water but soluble in organic solvents. Lipidomics refers to an extensive study of lipids and it targets how lipids are responsible for cell structure, cell signaling, and various other functions. Studies on lipids have proven that food intake and medicines can affect human bodies significantly and this pinpoints the importance of lipidomics. As advanced techniques and improvements in mass spectrometry and chromatography have expanded boundaries of lipidomics research field, lipid profiles have become one of the important aspects in work and our lab concentrates on analyzing lipids at a molecular level using various analyzing methods with high sensitivity.
Metabolites are directly or indirectly responsible for metabolic process, growth, and development, and affected by changes in external environments unlike DNA or proteins. They are mainly composed of lipids which are defined to be insoluble in water but soluble in organic solvents. Lipidomics refers to an extensive study of lipids and it targets how lipids are responsible for cell structure, cell signaling, and various other functions. Studies on lipids have proven that food intake and medicines can affect human bodies significantly and this pinpoints the importance of lipidomics. As advanced techniques and improvements in mass spectrometry and chromatography have expanded boundaries of lipidomics research field, lipid profiles have become one of the important aspects in work and our lab concentrates on analyzing lipids at a molecular level using various analyzing methods with high sensitivity.Use of nanoflow liquid chromatography maximizes desolvating effect, provides large area to volume ratios, and optimizes nebulization. By producing our own hand-made analytical columns, we can modify a length, diameter, packing materials, and other changes on columns depending on samples to be analyzed. Lipids can be separated by head groups in a normal-phase chromatography and by length of acyl chains in a reverse-phase chromatography. We analyze lipids in latter fashion using capillary columns which separates and elutes lipids according to the length of acyl chains and head groups simultaneously.
Two dimensional Separation of proteins by Isoelectric Focusing & multichannel Asymmetrical Flow Field-Flow Fractionation

Proteomics has captured the increased attention by providing insights to structures, functions, and changes in protein profiling of human body. High performance mass spectrometry can analyze proteins within a short period of time but as numerous types of proteins exist inside human body and their differences in concentrate can range up to 1010 folds, a separation science has become an important branch of proteomics as well. 2D-PAGE (PolyAcrylAmide Gel Electrophoresis) is upheld as one of the most general methods used for protein separation because of its great resolution for qualitative and quantitative purposes but it has its downsides; difficulty to systemize it automatically, relatively long analyzing time, and fluctuation of data. Our laboratory alternatively uses FFF, an elution technique based on a size separation, to prevent these problems and analyze the proteins using a shotgun method

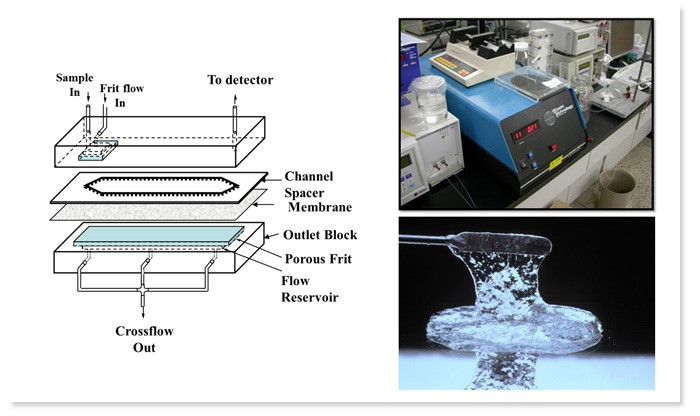
Characterization of hyaluronic acid using Frit Inlet Asymmetrical Flow Field-Flow Fractionzation & Multiangle Light Scattering

Hyaluronic acid (HA) is a water-soluble glycosaminoglycan with large molecular weight of 105 ~ 108 Da and is widely distributed in living organisms, from microbes to vertebrates. As HA is found in different types of tissues in the body, it is suitable to be used in cosmetic and biomedical applications such as eye surgery. The molecular weight and concentration of HA determine for which purpose HA will be used, so determination of exact molecular weight of HA is a critical step in both medical and cosmetic fields. Size exclusion chromatography is often utilized to determine the molecular weight but it risks sample contamination and sample might be absorbed to the packing materials.
Our laboratory developed Frit-Inlet Asymmetrical Flow Field-Flow Fractionation (FI-AFIFFF) using asymmetrical channel, which has a small sized frit in the sample inlet area of an acryl block in order to enable sample injection and separation simultaneously without applying focusing/relaxation process. FI-AFIFFF channel is easy to assemble and sample is unlikely to be absorbed to the channel membranes. Furthermore, by coupling it to Multiangle Light Scattering (MALS), an absolute quantification and characterization of the sample can be determined. Our laboratory is concentrating on analyzing pre-treated HAs using FFF, differential refractive index detector, and light scattering detector.
Our laboratory developed Frit-Inlet Asymmetrical Flow Field-Flow Fractionation (FI-AFIFFF) using asymmetrical channel, which has a small sized frit in the sample inlet area of an acryl block in order to enable sample injection and separation simultaneously without applying focusing/relaxation process. FI-AFIFFF channel is easy to assemble and sample is unlikely to be absorbed to the channel membranes. Furthermore, by coupling it to Multiangle Light Scattering (MALS), an absolute quantification and characterization of the sample can be determined. Our laboratory is concentrating on analyzing pre-treated HAs using FFF, differential refractive index detector, and light scattering detector.
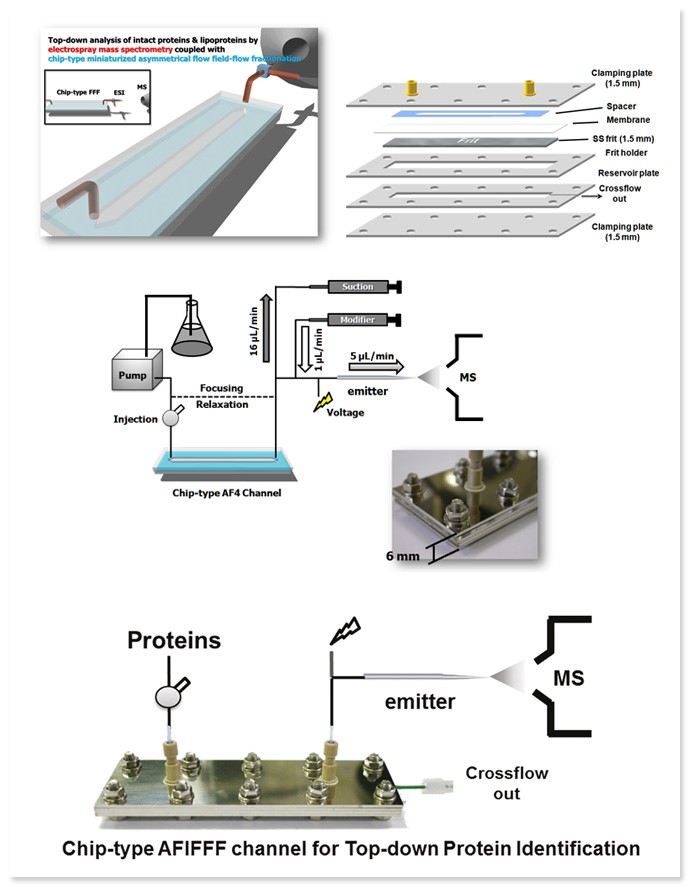
Analysis of proteins and lipids using Field-Flow Fractionation and Mass Spectrometry

We developed a chip-type miniaturized AFIFFF to analyze proteins by directly coupling it to electrospray ionization tandem mass spectrometry (ESI-MS). Constructed by stacking 1.5 mm thick stainless steel plates, the height of the channel became about 6 mm. Separation efficiency, detection limit, overloading effect, and reproducibility were tested by reducing the channel lengths while maintaining an equivalent flow rate and void time. Since the miniaturized AFIFFF operates at low flow rate with a MS-compatible buffer and removes salts and other impurities within the sample, it is suitable to couple AFIFFF with ESI-MS directly. Carbonic anhydrase (29 kDa) and transferrin from bovine serum (78 kDa) were used to confirm the separation and characterization of proteins were obtained by collision induced dissociation (CID) patterns. AFIFFF-ESI-MS is capable of detecting protein dimers unlike the conventional methods.